 
Another major usage of electron configuration can be seen in predicting the properties of an element group.It is useful in identifying the valency of elements.Some of the main uses of electronic configuration include: Look at the following examples for further understanding:Īluminium: 1 s 2 2 s 2 2 p 6 3 s 2 3 p 1 it becomes Ne 3 s 2 3 p 1Ĭalcium: 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 4 s 2 it becomes Ar 4 s 2 Hence, abbreviated sodium electron configuration is Ne 3 s 1 because Neon’s configuration is 1 s 2 2 s 2 2 p 6, which can be compressed to He 2 s 2 2 p 6. In those scenarios, a condensed or abbreviated notation will be highly helpful in naming the configuration rather than using the standard name.įurthermore, in the compressed notation, we can use the square brackets form for completely filled subshells while naming the noble gases. However, denoting the configuration based on standard notation will make the expression lengthy and complex, especially if an element contains a bigger atomic number. Here, the 1, in front of the s, denotes the energy level. With that, one can simply define the electronic configuration of an atom. An atomic subshell is nothing but the notation of the number of electrons they hold.įor instance, we can say the sodium electronic configuration as: 1s 2 2s 2 2p 6 3s 1 The basic arrangement of Electronic ConfigurationĬonfiguration of electrons involves three simple terms: Here, all the electrons that consist of atomic subshells are usually placed in a specific sequence. This configuration is capable of following a standard notation. Electron Configuration – DefinitionĪn element’s electron configuration illustrates how electrons can be distributed in atomic orbitals. This article will discuss how electrons are arranged and the electron configuration order in detail while having a brief look at other similar concepts. Such arrangements of electrons are termed electronic configuration. 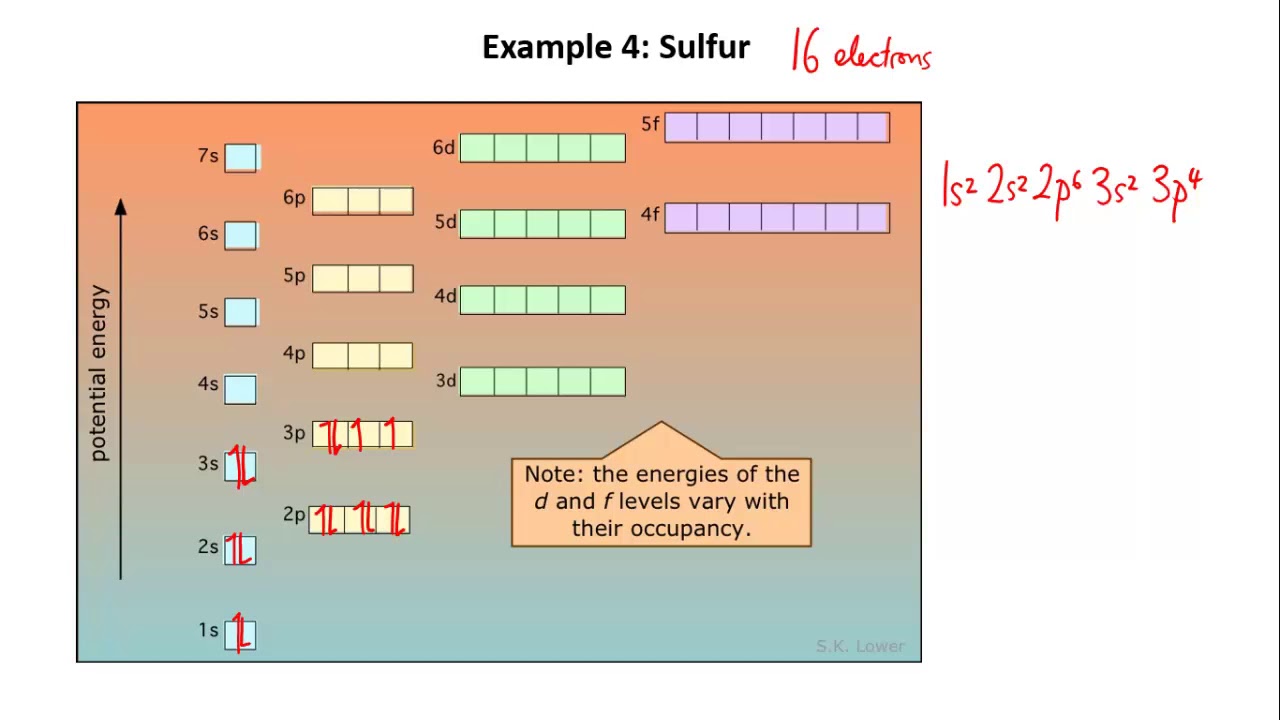
These electrons are arranged in a specific order inside the orbitals of the atoms. Electrons, negatively charged particles, are present on the outer shells of the atoms surrounding the nucleus. In that, protons are positively charged, whereas neutrons do not possess any charges. An atom contains a nucleus in its core, which is made up of protons and neutrons. Atomic structure is a fundamental topic in the field of Chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
